
Mathematics Behind pH Buffer Calculations
The Difference Between Thermodynamic pKa and Apparent pKa
A buffer species with a single protonated state is defined as
where is a weak acid, the conjugate base, and the charge of the conjugate base.
It follows that the dissociation constant is defined as
The dissociation constant, , is however more commonly presented as its own negative logarithm
Substituting and rearranging, we get the Henderson-Hasselbalch equation
However, this is only true under ideal conditions. In reality, many non-idealities exist. For example, any free ion would be shielded by oppositely charged ions in the solution. This changes the apparent concentration of . It can be said that the activity of changes. The activity is therefore defined as
where is the activity, is the activity coefficient, and is the concentration of , respectively.
Substituting the concentrations with the corresponding activities in equation (4), we get that
and thus
where is the apparent pKa under specific conditions, and is the thermodynamic pKa, determined at ideal conditions.
The activity coefficients vary widely for different species and under different conditions and must be determined experimentally.
How Temperature Influences pKa
The dissociation of a buffer species is an equilibrium process, and like all equilibria, it is affected by temperature. The enthalpy of dissociation, , governs how the equilibrium shifts as temperature changes. To a first-order approximation, this temperature dependence is linear and can be written as
where K (25°C) is the standard reference temperature at which values are typically reported, and is the temperature coefficient of the buffer species.
The magnitude and sign of varies considerably between buffers. Tris, for example, has a temperature coefficient of approximately K-1, meaning its pKa decreases by roughly 0.28 units for every 10°C increase. Phosphate, by contrast, has a coefficient of only K-1. This has important practical consequences: a Tris buffer prepared at 25°C and used at 4°C will have a pH approximately 0.6 units higher than intended, while a phosphate buffer under the same shift will change by less than 0.05 units.
How Ionic Strength Influences Apparent pKa
The presence of ions changes the activity of any acid-base pair. However, different acids are more affected than others.
The ionic strength of a solution is defined as
where is the concentration of ion , and is the charge of ion .
Empirical correlations between the activity of an acid-base pair and the ionic strength of the solution have been determined [1]. Substituting these into equation (7) and combining with the temperature correction from equation (8), we get the complete expression for the apparent pKa
where is the charge of the conjugate base and
and
where is the temperature in Kelvin and is the temperature in Celsius [2].
From the ionic strength term in equation (10), we see that for acids with a neutral or a positively charged conjugate base, the apparent pKa increases with increasing ionic strength. Conversely, for acids with a negatively charged conjugate base, the apparent pKa decreases with increasing ionic strength.
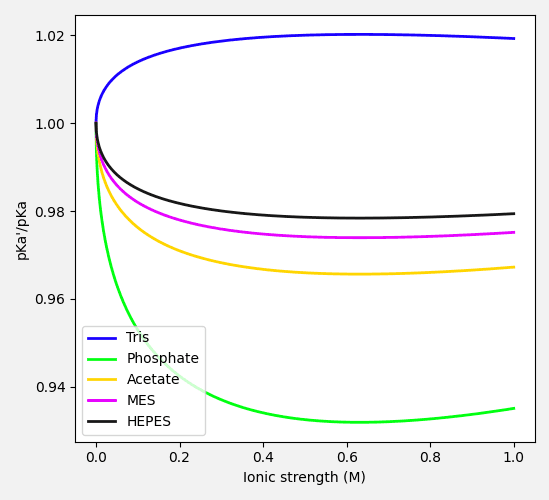
When calculating the apparent pKa in equation (10), it is important to recognize that the calculated apparent pKa affects the equilibrium ratio of the acidic and basic components of the buffer. This in turn leads to a change in total ionic strength of the solution, which subsequently affects the apparent pKa. It thus becomes an iterative problem to solve, with the calculated apparent pKa converging to a stable value after just a few iterations.
If you need to accurately calculate the recipe to create a buffer of a certain pH, taking ionic strength from salt additives and temperature into account, you can use our free-to-use buffer calculator.
References
Want to know more?
Efflux provides chromatography modeling and simulation software and services to leading biopharma companies.

